Introduction
Organization
Organization
Organization Introduction
Divisions
Seminar
Member
Education
Introduction
Organization
Organization
Organization Introduction
Divisions
Seminar
Member
Education


Organization
Organization Introduction
Divisions
Member
Education
Organization
Organization
Introduction
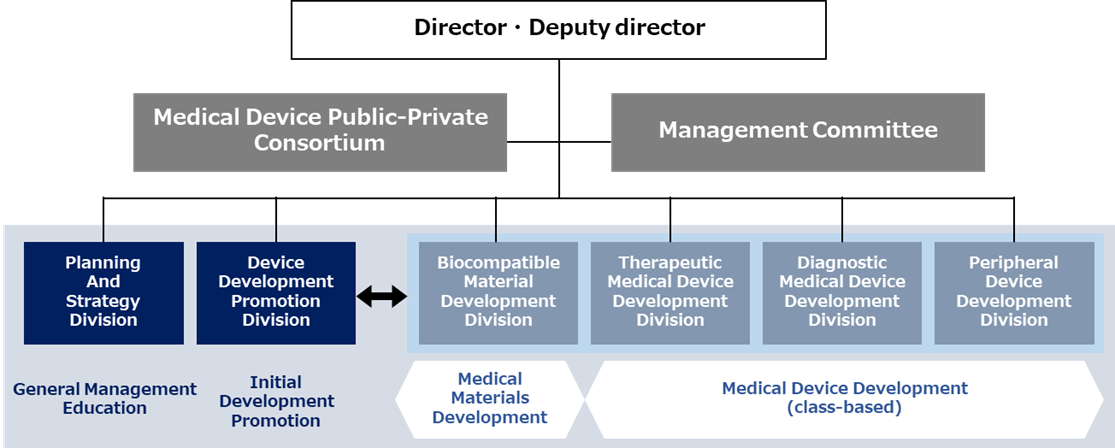
The Center has a Development Strategy Division that oversees the entire operation of the center and a Development Division for each class of medical device. The Pharmaceutical and Medical Device Law classifies medical devices into three categories, general medical devices, controlled medical devices, and highly controlled medical devices, based on the degree of risk they pose to the human body, with regulations varying according to this classification. These classifications have been taken into account in the structure of the division.

Director、Deputy director、Division Leader
Director
【Med】MURAGAKI Yoshihiro
Deputy director
【Med】FUKUMOTO Takumi
【Eng】MUKAI Toshiji
Planning And Strategy Division
【Eng】IMAI Yohsuke
【Med】TAKAO Toshitatsu
【Med】FUKUMOTO Takumi
【Med】KODAMA Yuuzou
【Med】SAKAI Yoshitada
【Med】ASARI Sadaki
【Med】HARA Yosuke
【Eng】MUKAI Toshiji
【Eng】ISONO Yoshitada
【Eng】YOKOKOHJI Yasuyoshi
【Eng】OZAWA Seiichi
Device Development Promotion Division
【Med】YASUDA Takahiro
【Eng】Nakadate Ryu
【Med】MURAGAKI Yoshihiro
【Med】MUNETO Koji
【Med】KUBOTA Fumio
【Med】OSHIYAMA Hiroakii
【Med】Fukuoka Yasushi
【Med】TOMIHATA Kenji
【Med】WADA Norihito
【Med】NISHIO Mizuho
【Med】YAMAGUCHI Tomoko
Biocompatible Material Development Division
【Eng】OOYA Tooru
【Med】MANIWA Yoshimasa
【Med】KURODA Ryosuke
【Med】TOYAMA Hirochika
【Eng】MUKAI Toshiji
【Eng】SHIOZAWA Daiki
【Eng】IKEO Naoko
Therapeutic Medical Device Development Division
【Eng】SUGANO Koji
【Med】YAMAGUCHI Raizou
【Med】NIBU Kenichi
【Med】KAKEJI Yoshihiro
【Med】OKADA Kenji
【Med】SASAKI Ryohei
【Med】SASAYAMA Takashi
【Med】UEMURA Munenori
【Med】UENO Hidetaka
【Eng】ISONO Yoshitada
【Eng】YOKOKOHJI Yasuyoshi
【Eng】MATSUYAMA Hideto
【Eng】NAGANO Hikaru
Diagnostic Medical Device Development Division
【Med】YAMAGUCHI Masato
【Eng】HAYASHI Kosuke
【Med】MURAKAMI Takamichi
【Med】MATSUOKA Hiroshi
【Med】KIDO Masahiro
【Med】FUKUZAWA Koji
【Med】OBATA Norihiko
【Eng】OHMURA Naoto
【Eng】IMAI Yohsuke
【Eng】MARUYAMA Tatsuo
【Eng】MISHIMA Tomokazu
【Eng】ISHIDA Shunichi
【Med】TAKEUCHI Toshifumi
Peripheral Device Development Division
【Med】BITO Yuko
【Med】KATO Hiroshi
【Eng】NISHIDA Isamu
Staff
【Eng】NAKATSUJI Tatsuya
Divisions

Planning And Strategy Division
Collaborate cross-functionally with each division of the center, formulate strategies for intellectual property, commercialization, pharmaceutical affairs, insurance coverage, etc., and develop products based on needs.

Device Development Promotion Division
Promote initial development to create medical device concepts based on clinical site needs, led by experienced developers, in collaboration with physicians and engineering researchers.

Biocompatible Material Development Division
Conduct development and evaluation of biocompatible materials required for implantable medical devices and needs assessment.

Therapeutic Medical Device Development Division
Conduct development and evaluation of Class III and IV highly controlled medical devices and needs assessment.

Diagnostic Medical Device Development Division
Conduct development and evaluation of Class II controlled medical devices and needs assessment.

Peripheral Device Development Division
Conduct development and evaluation of peripheral devices that do not fall under the category of medical devices, as well as needs assessment.
